
eBook - ePub
Transition Metal-Catalyzed Pyridine Synthesis
Transition Metal-Catalyzed Heterocycle Synthesis Series
Xiao-Feng Wu
This is a test
- 90 Seiten
- English
- ePUB (handyfreundlich)
- Über iOS und Android verfügbar
eBook - ePub
Transition Metal-Catalyzed Pyridine Synthesis
Transition Metal-Catalyzed Heterocycle Synthesis Series
Xiao-Feng Wu
Angaben zum Buch
Buchvorschau
Inhaltsverzeichnis
Quellenangaben
Über dieses Buch
Transition Metal-Catalyzed Pyridine Synthesis provides an overview of pyridines, describing properties of these heterocycle compounds and describing traditional synthetic procedures for them. The book then explores catalyzed procedures for pyridine synthesis in greater detail and depth than is currently available in published Review articles.
The short series Transition Metal-Catalyzed Heterocycles Synthesis, authored by Xiao-Feng Wu, summarizes recent achievements on heterocycles synthesis with transition metal as the catalysts, with each volume dedicated to one heterocycle compound.
- Brief, focused review of this active research area, Pyridine synthesis via transition metal catalysis
- Useful coverage of Pyridine properties and both intermolecular and intramolecular cyclization
- Volume Two in Elsevier's short work series, "Transition Metal-Catalyzed Heterocycles Synthesis"
Häufig gestellte Fragen
Wie kann ich mein Abo kündigen?
Gehe einfach zum Kontobereich in den Einstellungen und klicke auf „Abo kündigen“ – ganz einfach. Nachdem du gekündigt hast, bleibt deine Mitgliedschaft für den verbleibenden Abozeitraum, den du bereits bezahlt hast, aktiv. Mehr Informationen hier.
(Wie) Kann ich Bücher herunterladen?
Derzeit stehen all unsere auf Mobilgeräte reagierenden ePub-Bücher zum Download über die App zur Verfügung. Die meisten unserer PDFs stehen ebenfalls zum Download bereit; wir arbeiten daran, auch die übrigen PDFs zum Download anzubieten, bei denen dies aktuell noch nicht möglich ist. Weitere Informationen hier.
Welcher Unterschied besteht bei den Preisen zwischen den Aboplänen?
Mit beiden Aboplänen erhältst du vollen Zugang zur Bibliothek und allen Funktionen von Perlego. Die einzigen Unterschiede bestehen im Preis und dem Abozeitraum: Mit dem Jahresabo sparst du auf 12 Monate gerechnet im Vergleich zum Monatsabo rund 30 %.
Was ist Perlego?
Wir sind ein Online-Abodienst für Lehrbücher, bei dem du für weniger als den Preis eines einzelnen Buches pro Monat Zugang zu einer ganzen Online-Bibliothek erhältst. Mit über 1 Million Büchern zu über 1.000 verschiedenen Themen haben wir bestimmt alles, was du brauchst! Weitere Informationen hier.
Unterstützt Perlego Text-zu-Sprache?
Achte auf das Symbol zum Vorlesen in deinem nächsten Buch, um zu sehen, ob du es dir auch anhören kannst. Bei diesem Tool wird dir Text laut vorgelesen, wobei der Text beim Vorlesen auch grafisch hervorgehoben wird. Du kannst das Vorlesen jederzeit anhalten, beschleunigen und verlangsamen. Weitere Informationen hier.
Ist Transition Metal-Catalyzed Pyridine Synthesis als Online-PDF/ePub verfügbar?
Ja, du hast Zugang zu Transition Metal-Catalyzed Pyridine Synthesis von Xiao-Feng Wu im PDF- und/oder ePub-Format sowie zu anderen beliebten Büchern aus Physical Sciences & Organic Chemistry. Aus unserem Katalog stehen dir über 1 Million Bücher zur Verfügung.
Information
Thema
Physical SciencesThema
Organic ChemistryChapter 1
Introduction
Abstract
A short introduction on pyridine derivative has been given.
Keywords
Pyridine; biological active; organic synthesis
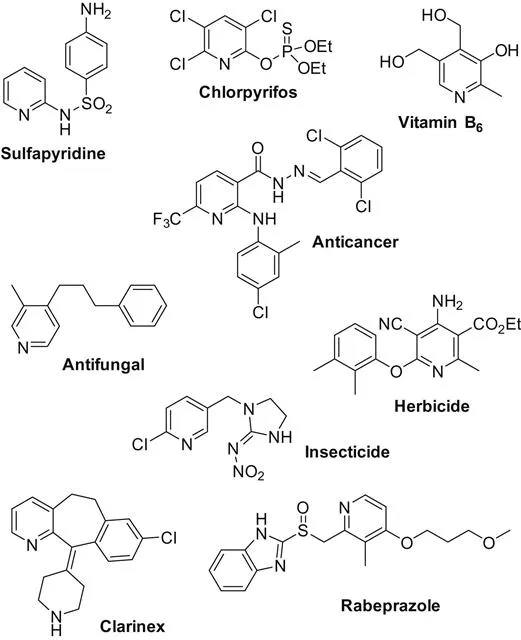
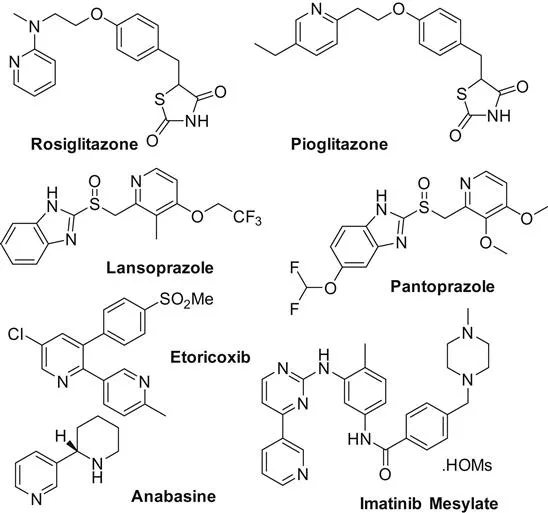
Pyridine is an important class of nitrogen-containing heterocycles found in various natural products, pharmaceuticals, and materials (Scheme 1.1). Based on its importance, numerous synthetic procedures have been developed for their preparation [1]. In this book volume, the main achievements on transition metal-catalyzed pyridines synthesis are discussed. Based on the reaction types, the whole volume is catalogued by intramolecular cyclization and intermolecular cyclization reactions.
Chapter 2
Synthesized by Intramolecular Cyclizations
Abstract
The procedures based on transition metal-catalyzed intramolecular cyclizations to prepare pyridines have been discussed here.
Keywords
Pyridine; intramolecular cyclization; organic synthesis; synthetic methodology; carbonylation; coupling
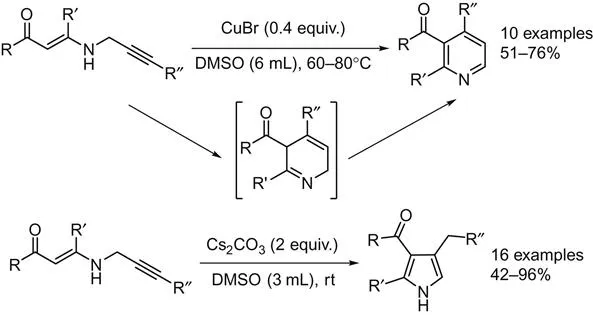
Intramolecular cyclization of substrates to the corresponding pyridine derivatives is the most straightforward pathway. By intramolecular version reaction, the positions of substituents are fixed and easier to control, such as N-propargylic β-enaminones in pyridines and pyrroles synthesis. In 2008, Cacchi et al. reported a copper-catalyzed synthesis of polysubstituted pyridines from N-propargylic β-enaminones [1]. By using DMSO as the solvent, pyrroles can be produced in good to high yields in the presence of only Cs2CO3 at room temperature, while pyridines can be formed under the assistant of CuBr at 60–80°C (Scheme 2.1). Here, the N-propargylic β-enaminones can be prepared via the following sequences: (1) cross-coupling of terminal alkynes with acyl chlorides; (2) followed by the conjugate addition of propargylamine with the resultant α,β-enones; (3) further Sonogashira cross-coupling of the propargyl derivative with aryl halides.
A procedure by using enamino ester and alkynone as the substrates was developed as well [2]. 2,3,4,6-Tetrasubstituted pyridines were prepared in a single step. Various acids, such as acetic acid, Amberlyst 15 ion exchange resin, zinc(II) bromide or ytterbium(III) triflate, can be applied as promoter for the cyclization step of the Michael addition adduct. 4-(3-Oxoalkyl)isoxazoles found could be applied as starting material for pyridine synthesis as well [3].
In 2013, Kim and coworkers reported a palladium-catalyzed domino cyclization of N-(2-bromoallyl)-N-cinnamyltosylamides for the construction of pyridines [4]. The reaction proceeds via a domino 5-exo/3-exo carbopalladation, ring expansion by palladium rearrangement, and an aromatization. Various 4-arylnicotinate derivatives were produced in good yields (Scheme 2.2).
Inhaltsverzeichnis
- Cover image
- Title page
- Table of Contents
- Copyright
- Dedication
- Chapter 1. Introduction
- Chapter 2. Synthesized by Intramolecular Cyclizations
- Chapter 3. Synthesized by Intermolecular Cyclizations
- Chapter 4. Summary and Outlook
Zitierstile für Transition Metal-Catalyzed Pyridine Synthesis
APA 6 Citation
Xiao-Feng. (2016). Transition Metal-Catalyzed Pyridine Synthesis ([edition unavailable]). Elsevier Science. Retrieved from https://www.perlego.com/book/1830185/transition-metalcatalyzed-pyridine-synthesis-transition-metalcatalyzed-heterocycle-synthesis-series-pdf (Original work published 2016)
Chicago Citation
Xiao-Feng. (2016) 2016. Transition Metal-Catalyzed Pyridine Synthesis. [Edition unavailable]. Elsevier Science. https://www.perlego.com/book/1830185/transition-metalcatalyzed-pyridine-synthesis-transition-metalcatalyzed-heterocycle-synthesis-series-pdf.
Harvard Citation
Xiao-Feng (2016) Transition Metal-Catalyzed Pyridine Synthesis. [edition unavailable]. Elsevier Science. Available at: https://www.perlego.com/book/1830185/transition-metalcatalyzed-pyridine-synthesis-transition-metalcatalyzed-heterocycle-synthesis-series-pdf (Accessed: 15 October 2022).
MLA 7 Citation
Xiao-Feng. Transition Metal-Catalyzed Pyridine Synthesis. [edition unavailable]. Elsevier Science, 2016. Web. 15 Oct. 2022.